Why Surface Disinfection Matters in a Resistant World
By Sarah C. Bell-West, Ph.D., Senior Scientist, Clorox Healthcare
This article originally appeared in the APUA Newsletter, Volume 33, No. 3.
 Resistance to antimicrobial agents, especially antibiotics, is a major public health concern throughout the world. In the United States alone, The Centers for Disease Control and Prevention (CDC) estimates that antibiotic-resistant bacteria cause two million illnesses and approximately 23,000 deaths each year.1 Multimodal infection prevention approaches coupled with antimicrobial stewardship efforts are essential for the prevention of infections caused by antimicrobial resistant organisms. A recent CDC report estimated that if improved infection control practices and antibiotic stewardship efforts were adopted nationally, 619,000 infections and 37,000 deaths could be prevented over five years.2
Resistance to antimicrobial agents, especially antibiotics, is a major public health concern throughout the world. In the United States alone, The Centers for Disease Control and Prevention (CDC) estimates that antibiotic-resistant bacteria cause two million illnesses and approximately 23,000 deaths each year.1 Multimodal infection prevention approaches coupled with antimicrobial stewardship efforts are essential for the prevention of infections caused by antimicrobial resistant organisms. A recent CDC report estimated that if improved infection control practices and antibiotic stewardship efforts were adopted nationally, 619,000 infections and 37,000 deaths could be prevented over five years.2
Given the growing problem of antibiotic resistance, environmental cleaning and disinfection can play an important role in helping to prevent the spread of antibiotic-resistant bacteria such as methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), carbapenem-resistant Enterobacteriaceae (CRE) in addition to other deadly pathogens associated with antibiotic usage, including Clostridium difficile. Additionally, the infection control community is asking questions about the relationship between disinfectant use and antibiotic resistance.
This article will focus on the role of surface disinfection in reducing transmission of antibiotic-resistant bacteria and the relationship between antibiotic resistance and the susceptibility of bacteria to surface disinfectants.
Does surface disinfection help to reduce transmission of antibiotic resistant bacteria?
Compliant cleaning and disinfection of environmental surfaces and medical equipment is a critical first line of defense against all pathogens in the healthcare environment, and especially those that are resistant to antibiotics. There is a growing body of evidence to demonstrate the effectiveness of environmental cleaning and disinfection as part of multimodal strategies to reduce pathogen transmission, prevent healthcare associated infections (HAIs), and improve patient outcomes.3 For example, researchers in Canada recently demonstrated that compliant daily cleaning and disinfection of high-touch surfaces in patient rooms (>80 percent cleaning compliance) was associated with a significant reduction in infections caused by MRSA, VRE, and Clostridium difficile in their hospital.4
However, studies have also demonstrated that often times fewer than 50 percent of hospital room surfaces are adequately cleaned and disinfected, potentially putting patients at increased risk for acquiring an HAI.5 This was documented in a recently published review and meta-analysis of six studies highlighting that a patient in a room that previously housed a patient colonized or infected with a pathogenic bacteria, which included MRSA, VRE, or Clostridium difficile, was twice as likely to be infected with the same pathogen.6 Antibiotic-resistant bacteria are not always the most common or deadly, underscoring the need for infection prevention strategies to focus on all HAI-causing pathogens, regardless of their antibiotic susceptibility. This was highlighted by a recent study in infant populations showing methicillin susceptible Staphylococcus aureus (MSSA) infections were more common and more deadly than those caused by MRSA.7
Does the use of surface disinfectants contribute to an increased risk for antimicrobial resistance?
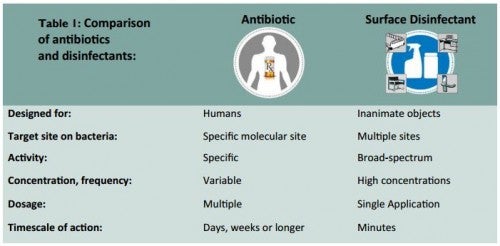
The topic of whether there is a relationship between the use of surface disinfectants in the home and in healthcare settings and the development and selection of antibiotic resistant organisms is heavily debated within the field of infection control.8 Antibiotics and surface disinfectants differ in both their mode and speed of action (Table 1). Antibiotics often work through a specific site of action, and bacteria can adapt to overcome the action of the drug over time. Surface disinfectants often have multiple, non-specific target sites across different classes of microorganisms. For example, oxidative disinfectants like sodium hypochlorite and hydrogen peroxide quickly and indiscriminately react with proteins, nucleic acids and other biomolecules, leading to oxidative cell destruction. This activity makes it challenging for pathogens to develop mechanisms to survive exposure to these surface disinfectants.
There is emerging research that suggests that the use of quaternary ammonium-based antimicrobial compounds may lead to decreased susceptibility in bacteria and cross-cross resistance to antibiotics.9 Studies have demonstrated that exposure of bacteria to sublethal concentrations of quaternary ammonium compounds in laboratory settings, which mimics disinfectant misuse over long time periods, can trigger resistance mechanisms.10 There is also evidence to support the position that the use of disinfectants does not contribute to antibiotic resistance, and some have cited studies demonstrating that both antibiotic resistant and antibiotic susceptible bacteria are still susceptible to surface disinfectants under normal usage conditions.11, 12 For example, Campos and colleagues in Brazil recently reported that there was no significant difference in the susceptibility of clinical isolates MRSA and MSSA to various disinfection chemistries.13 While there is evidence to support both viewpoints, additional research is required to understand the relationship between sustained usage of antiseptics and disinfectants in clinical settings and the corresponding changes in antimicrobial susceptibility of clinical isolates over time.
Where do we go from here?
A multi-faceted approach utilizing both infection control and antimicrobial stewardship strategies is needed to reduce the infection burden attributed to antimicrobial resistant organisms. Killing pathogenic bacteria, viruses and fungi on surfaces before they infect a host can reduce the number of difficult-to-treat infections caused by antibiotic resistant organisms and reduce the risk of the development of antibiotic resistance. Combining the key principles of antimicrobial stewardship14 to include proper selection and usage of surface disinfectants,15 the following steps are recommended:
- Educate all staff, including clinical and environmental services team members, about the burden of drug-resistant infections in your facility.
- Communicate internally when drug-resistant infections in patients are identified and externally when patient with a drug-resistant infection are transferred to another facility.
- Protect patients from drug-resistant infections by implementing best practices in infection control.
- Reinforce the importance of compliant cleaning and disinfection of medical equipment as well as hard and soft environmental surfaces to reduce pathogen contamination of surfaces prevent pathogen transmission.
- Follow relevant guidelines and precautions at every patient encounter.
- Prescribe antibiotics wisely.
- Remove temporary medical devices such as catheters and ventilators as soon as they are no longer needed.
References
- Centers for Disease Control and Prevention (CDC). Antibiotic / Antimicrobial Resistance. http://www.cdc.gov/ drugresistance/ (accessed Nov 23, 2015).
- Slayton RB, Toth D, Lee BY, et al. Vital Signs: Estimated Effects of a Coordinated Approach for Action to Reduce Antibiotic-Resistant Infections in Health Care Facilities – United States. MMWR. Morb. Mortal. Wkly. Rep. 2015, 64 (30), 826–831.
- Han JH, Sullivan N, Leas BF, et al. Cleaning Hospital Room Surfaces to Prevent Health Care-Associated Infections: A Technical Brief. Ann. Intern. Med. 2015, 163 (8), 598–607 DOI: 10.7326/M15-1192.
- Alfa MJ, Lo E, Olson N, MacRae M, Buelow-Smith L. Use of a daily disinfectant cleaner instead of a daily cleaner reduced hospital-acquired infection rates. Am. J. Infect. Control 2015, 43 (2), 141–146 DOI: 10.1016/ j.ajic.2014.10.016.
- Carling PC, Huang SS, Improving healthcare environmental cleaning and disinfection: current and evolving issues. Infect. Control Hosp. Epidemiol. 2013, 34 (5), 507–513 DOI: 10.1086/670222.
- Mitchell BG, Dancer SJ, Anderson M, Dehn E. Risk of organism acquisition from prior room occupants: a systematic review and meta-analysis. J. Hosp. Infect. 2015, 91 (3), 211–217 DOI: 10.1016/j.jhin.2015.08.005.
- Ericson JE, Popoola VO, Smith PB et al. Burden of Invasive Staphylococcus aureus Infections in Hospitalized Infants. JAMA Pediatr. 2015, 1 DOI: 10.1001/jamapediatrics.2015.2380.
- Harbarth S, Tuan Soh S, Horner C, Wilcox MH. Is reduced susceptibility to disinfectants and antiseptics a risk in healthcare settings? A point/counterpoint review. J. Hosp. Infect. 2014, 87 (4), 194–202 DOI: 10.1016/ j.jhin.2014.04.012.
- Gerba CP. Quaternary ammonium biocides: efficacy in application. Appl. Environ. Microbiol. 2015, 81 (2), 464 –469 DOI: 10.1128/AEM.02633-14.
- Buffet-Bataillon S, Tattevin P, Bonnaure-Mallet M, Jolivet-Gougeon A. Emergence of resistance to antibacterial agents: the role of quaternary ammonium compounds–a critical review. Int. J. Antimicrob. Agents 2012, 39 (5), 381–389 DOI: 10.1016/ j.ijantimicag.2012.01.011.
- Weber DJ, Rutala WA. Use of germicides in the home and the healthcare setting: is there a relationship between germicide use and antibiotic resistance? Infect. Control Hosp. Epidemiol. 2006, 27 (10), 1107–1119 DOI: 10.1086/507964.
- Weber, D J, Rutala, WA, Sickbert-Bennett, EE, Outbreaks associated with contaminated antiseptics and disinfectants. Antimicrob. Agents Chemother. 2007, 51 (12), 4217–4224 DOI: 10.1128/AAC.00138-07.
- Campos GB, Souza SG, Lob O, et al. Isolation, molecular characteristics and disinfection of methicillin -resistant Staphylococcus aureus from ICU units in Brazil. New Microbiol. 2012, 35 (2), 183–190.
- Centers for Disease Control and Prevention (CDC). Core Elements of Hospital Antibiotic Stewardship Programs -Get Smart for Healthcare. http://www.cdc.gov/ getsmart/healthcare/implementation/core-elements.html (accessed Nov 25, 2015).
- Rutala WA, Weber DJ. Selection of the ideal disinfectant. Infect. Control Hosp. Epidemiol. 2014, 35 (7), 855–865 DOI: 10.1086/676877.




